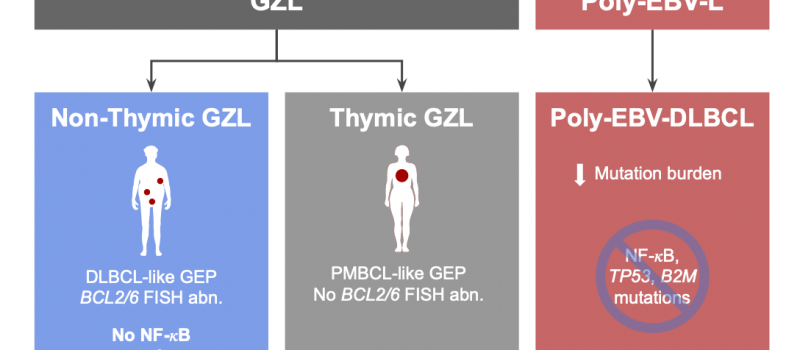
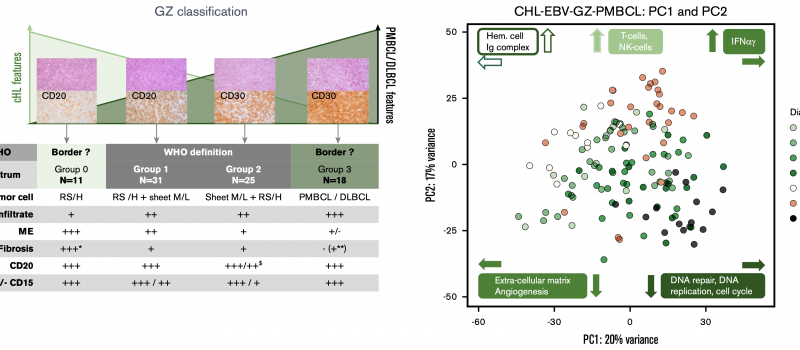
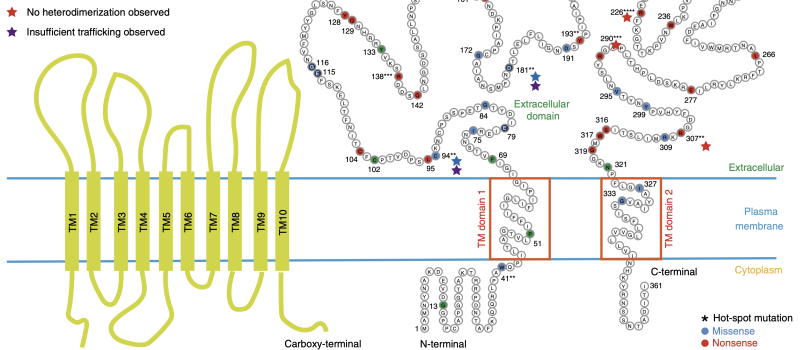
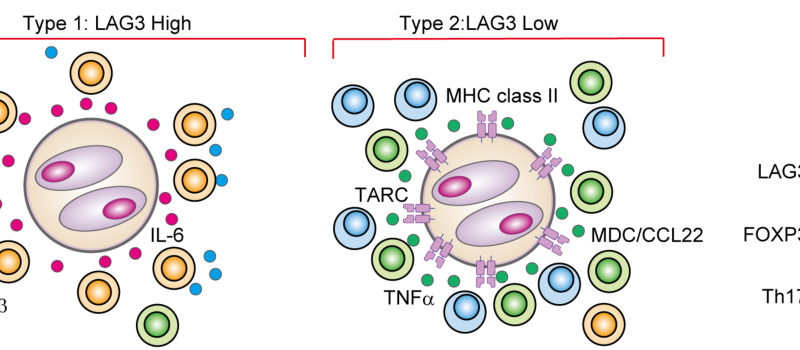
Welcome to the Steidl Lab!
The research of the Steidl laboratory focuses on the molecular characterization of lymphoid cancers and biomarker discovery in a variety of Hodgkin and Non-Hodgkin lymphoma subtypes. Our translational lymphoma research group uses a variety of state-of-the-art genome-wide discovery tools including array-based and digital gene expression profiling, single-nucleotide polymorphisms analysis and massively parallel sequencing techniques. A main research goal of the lab is to identify and functionally study underlying genetic mechanisms of immune privilege in lymphoid cancers. In previous work using next generation sequencing, we identified several driver mutations and gene fusions, in particular, that have been shown to arise from specific chromosomal rearrangements in Hodgkin lymphoma and primary mediastinal large B cell lymphoma. Most of the identified genes (e.g. CIITA, CD274, PDCD1LG2) feature prominently in immune cell function impacting non-neoplastic cells in the tumour microenvironment. The discovery of these changes across a wide spectrum of lymphomas will likely reinforced the paradigm that immune privilege is a critical component of cancer phenotypes.
Another focus of the laboratory is on the specific composition of the tumour microenvironment in Hodgkin lymphoma. Using gene expression profiling and immunohistochemistry we have identified that the number of tumour-associated macrophages in lymph node biopsies is linked to unfavourable treatment outcome. However, many questions remain regarding the molecular mechanisms underlying the interaction of the malignant cells with infiltrating macrophages and immune cells in the microenvironment in general. Recently, CSF1R has been identified as a key molecule expressed on Hodgkin Reed Sternberg (HRS) cells and tumour-associated macrophages in Hodgkin lymphoma. We now seek to better describe the phenotype of tumour-associated macrophages found in lymphoma biopsies to shed more light on this tumour-microenvironment interaction and to test if this cross-talk can be targeted by small molecule inhibitors in-vitro.
Lymphoma relapse and progression is still one of the major clinical challenges in clinical care and our knowledge about the underlying biology of relapse is still rudimentary. Our preliminary data indicate that certain gene findings have developed during treatment and might be characteristic of this relapse biology. Therefore, we specifically focus in ongoing translational studies on lymphoma relapse biopsies using highly-annotated clinical data sets. By collaboration with our clinical colleagues we are working on solutions how to develop better outcome predictors and predictive biomarkers that inform on individualized treatment options. These studies encompass biomarker evaluation in Hodgkin lymphoma, follicular lymphoma, large B cell lymphomas, and mantle cell lymphoma.
Collaborations
As a translational research laboratory we pursue the goal of translating our research findings into better treatments for our patients. The Centre for Lymphoid Cancer (CLC) provides an excellent infrastructure to answer clinically relevant questions in collaboration with clinical oncologists (Joseph Connors, Laurie Sehn, Kerry Savage), hemato-pathologists (Randy Gascoyne), genome researchers (Marco Marra) and computational biologists (Sohrab Shah). The Centre for Translational and Applied Genomics (CTAG) and the Genome Sciences Centre (GSC) is providing cutting technology to facilitate translational research from bench to bedside. Multiple collaborations with researchers inside and outside the Cancer Agency complement the research focus areas to provide additional expertise in epigenomics (Martin Hirst) in vivo modelling (Steidl lab, Einstein College, NY), endogenous retroviruses and epigenetics (Mager lab, Terry-Fox Laboratory, BCCA), immune pathology (Anke van den Berg, Arjan Diepstra, University of Groningen, NL), and childhood lymphomas (Horton lab, Baylor College, Houston, TX).